
Lewis dot structure of CH3OH Quizlet
An isolated carbon owns 4 valence electrons. The bound carbon in methanol owns (½ x 8) = 4 valence electrons: formal charge on carbon =. (4 valence electron on isolated atom) - (0 nonbonding electrons) - (½ x 8 bonding electrons) = 4 - 0 - 4 = 0. So the formal charge on carbon is zero. For each of the hydrogens in methanol, we also get a.
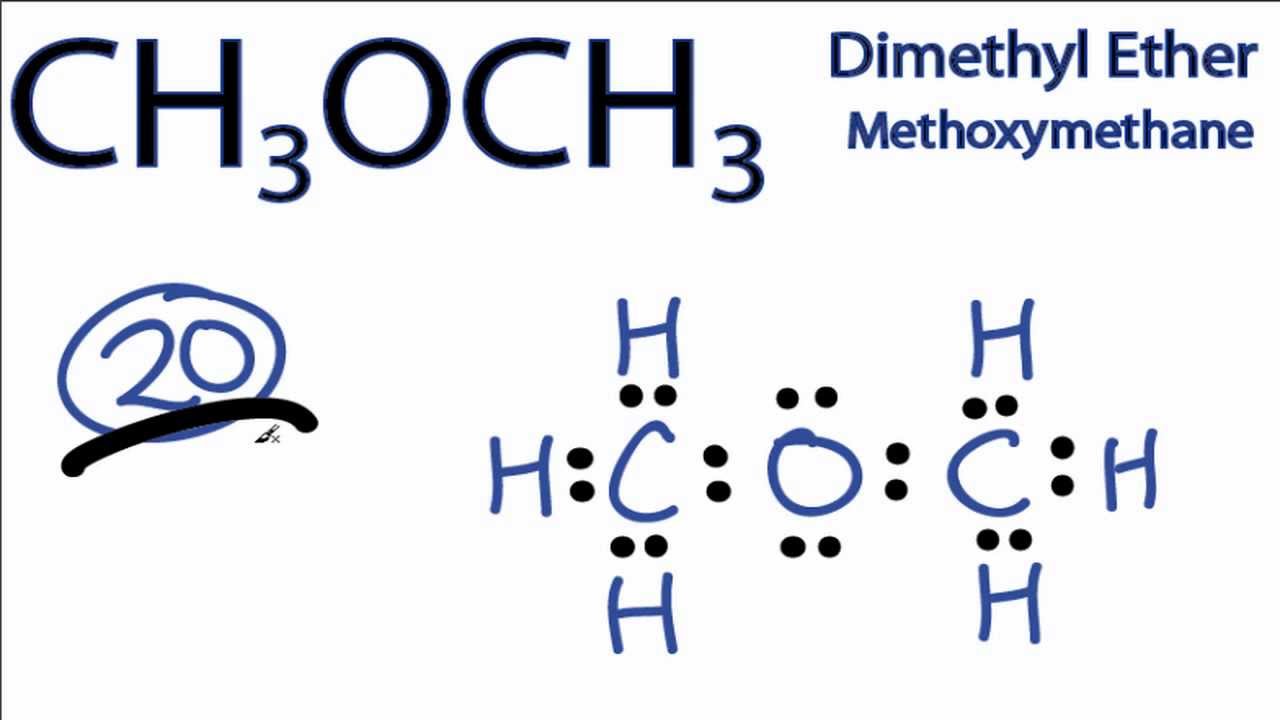
CH3OCH3 Lewis Structure How to Draw the Lewis Structure for CH3OCH3
May 22, 2023 by Jay Rana Ready to learn how to draw the lewis structure of CH3OH? Awesome! Here, I have explained 6 simple steps to draw the lewis dot structure of CH3OH (along with images). So, if you are ready to go with these 6 simple steps, then let's dive right into it!

i. What is the Lewis structure for CH3CHCHOH? (If resonance is present
How to draw lewis structure of CH3OH? The Lewis structure of methanol (CH3OH) consists of a carbon (C) atom at the center which is bonded to three atoms of hydrogen (H) and a hydroxyl (OH) functional group. In this way, there are a total of 4 electron density regions around the central C-atom in the CH3OH Lewis structure.

Solved Draw the product formed when the structure shown
Methanol (CH 3 OH) is the simplest alcohol which has only one carbon atom. According to the lewis structure of methanol, it has one O-H bond, three C-H bonds and one C-O bond. There are 2 lone pairs on oxygen atom. There are total of 14 electrons in valence shells in the overall molecule as lone pairs and bonds. CH 3 OH lewis structure

How to Draw the Lewis Structure for CH3OH (Methanol) YouTube
1.93K subscribers Subscribe 2K views 1 year ago Lewis Structure Hey Guys! For today's video, we are going to study the Lewis dot structure of Methanol. It has a c Lewis Structure of SO4.
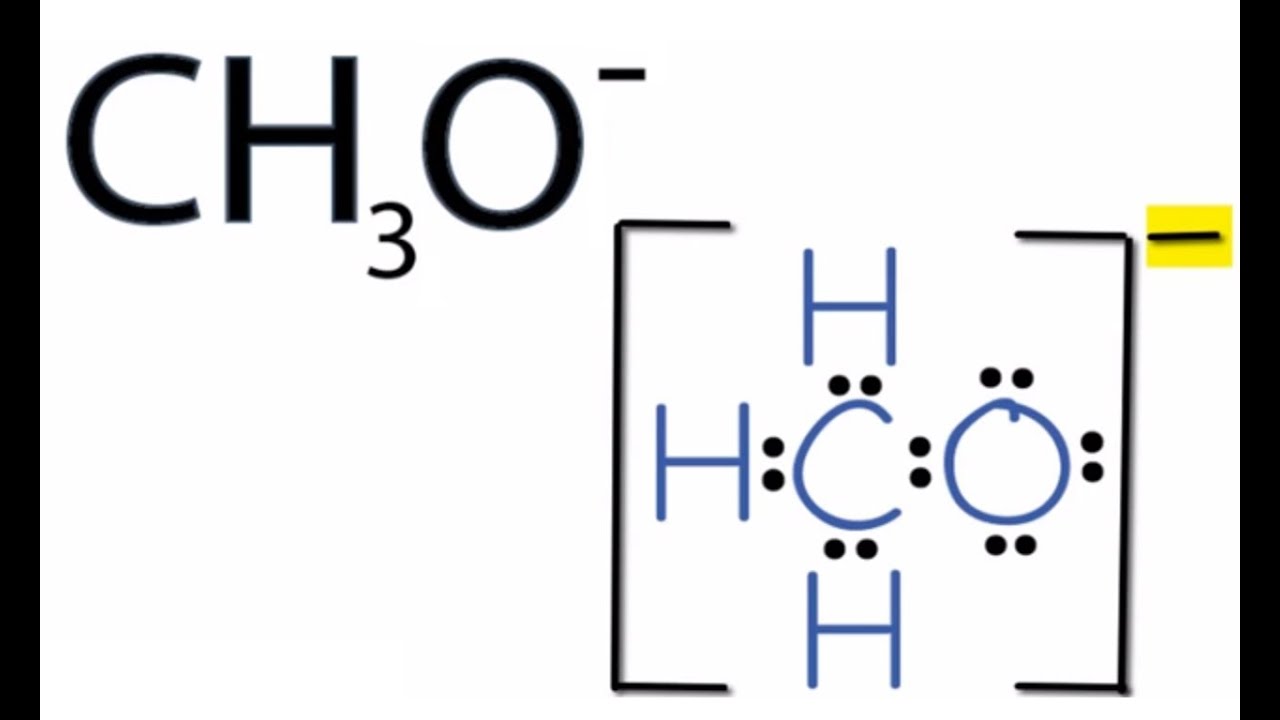
CH3O Lewis Structure How to Draw the Lewis Structure for CH3O YouTube
A step-by-step explanation of how to draw the CH3OH Lewis Structure.When you see a carbon with an OH attached (like CH3OH, C2H5OH, etc.) that means that he O.
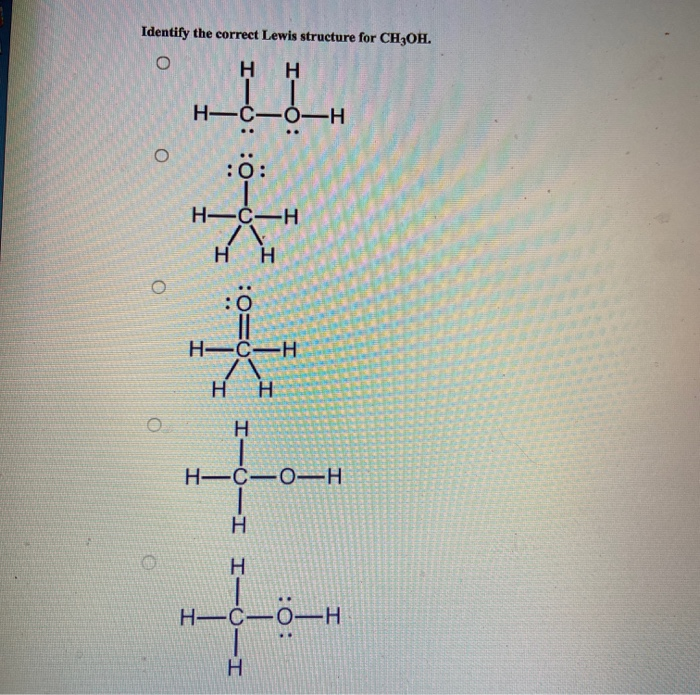
Solved Identify the correct Lewis structure for CH3OH. o H H
Lewis Dot Structure of CH3OH (Methanol) kentchemistry.com 25.1K subscribers Subscribe Subscribed 127K views 12 years ago Every Video I quickly take you through how to draw the Lewis.
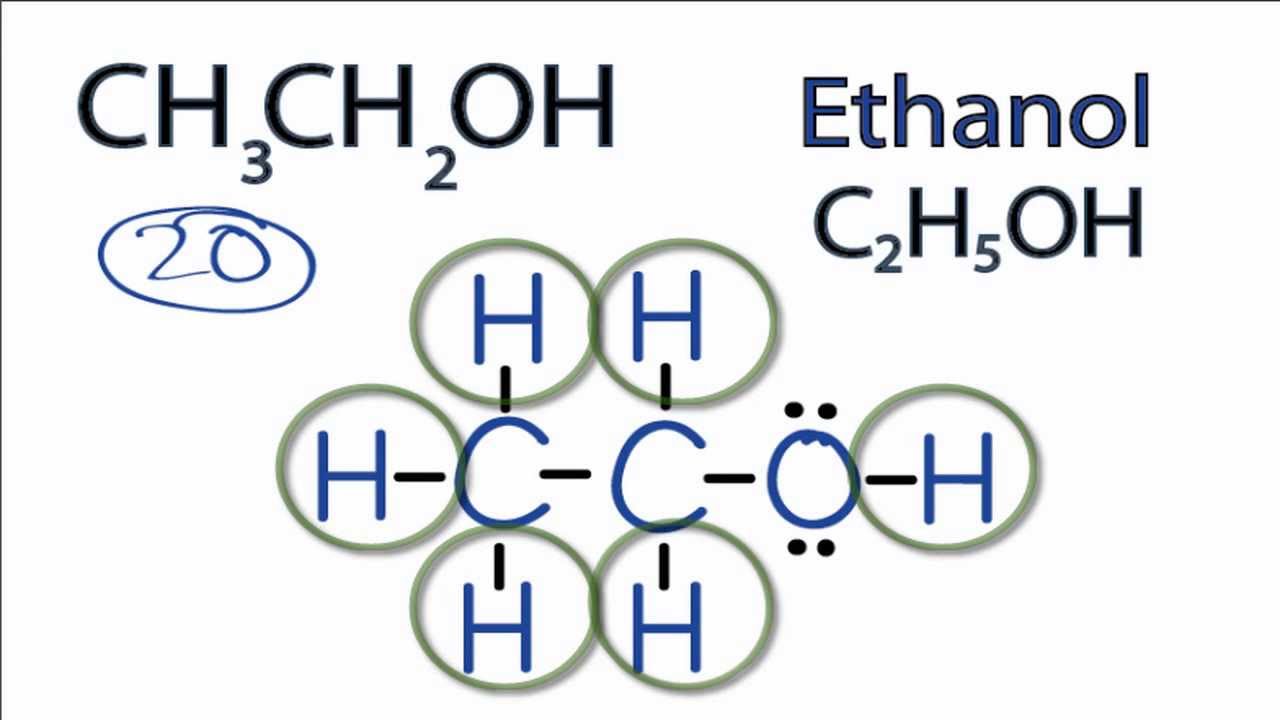
Lewis Structure For C2h5oh
This widget gets the Lewis structure of chemical compounds. Send feedback | Visit Wolfram|Alpha Get the free "Lewis Structure Finder" widget for your website, blog, Wordpress, Blogger, or iGoogle. Find more Chemistry widgets in Wolfram|Alpha.
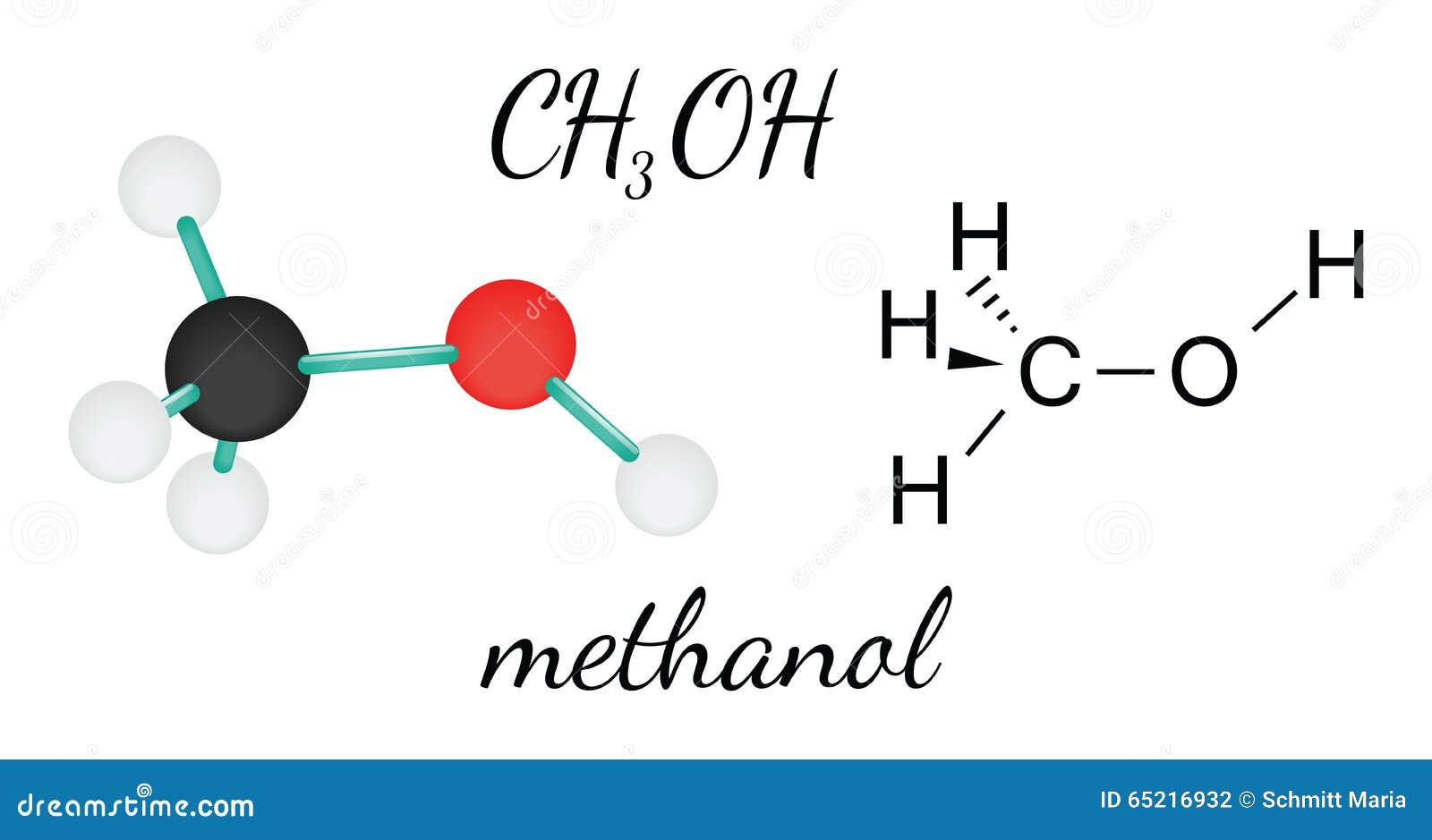
CH3OH methanol molecule stock vector. Illustration of hydrogen 65216932
Lewis dot structure is a pictorial representation of the molecule, it's bonding with other atoms and the arrangement of atoms in the compound. It helps in knowing the number of bonded electrons, lone pairs, and the compound's molecular shape.

What type of intermolecular forces are Persent in CH3OH?
Step 1: Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. When drawing the structure of an ion, be sure to add/subtract electrons to account for the charge. Step 2: Connect the atoms to each other with single bonds to form a "skeleton structure.".
CH3OH Lewis Structure How to Draw the Lewis Structure for CH3OH
Watch on Steps of drawing CH3OH lewis structure Step 1: Find the total valence electrons in CH3OH molecule In order to find the total valence electrons in CH3OH molecule, first of all you should know the valence electrons present in carbon atom, hydrogen atom as well as oxygen atom.

Spice of Lyfe Methanol Chemical Equation
A step-by-step explanation of how to draw the CH3O- Lewis Dot Structure.Note that you should put the CH3O- Lewis structure in brackets with as 1- on the outs.

Estructura De Lewis Ch3oh Compuesto
The Lewis structure of CH3OH, also known as methanol, is a representation of the molecule's bonding and electron distribution. It provides valuable insights into the molecule's geometry, hybridization, and polarity. Let's explore the step-by-step process of determining the Lewis structure of CH3OH. Calculation of Valence Electrons

Hybridization of CH3OH YouTube
The Lewis Structure of a molecule gives the simplest representation of valence shell electrons around itself. Here, the valence electrons are represented by small dots and since a single bond consists of two bonding electrons, the two dots between two atoms are represented by a line instead, which represents a bond between them.
Ch3oh
We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 7.9 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 7.9 Lewis symbols illustrating the number of.
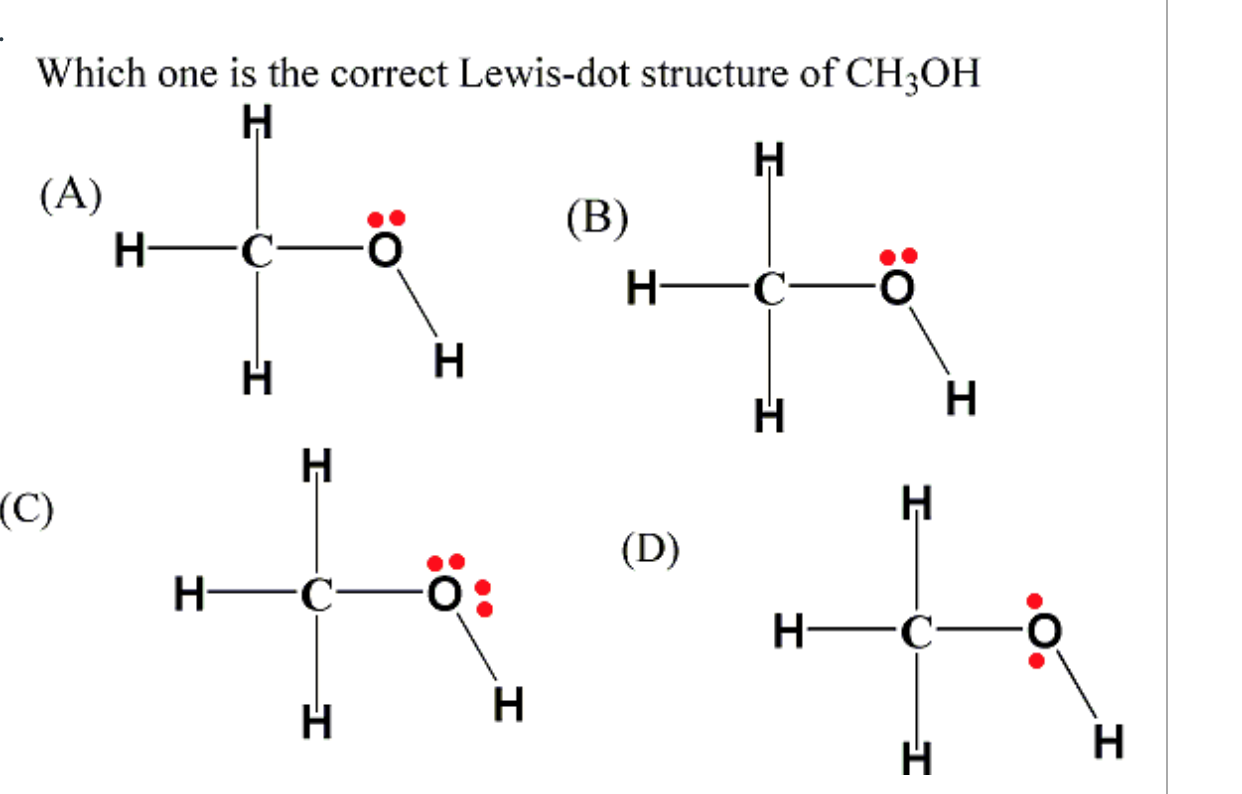
Ch3Oh Lewis Structure
The molecule methanol (methyl alcohol) has the structure CH3OH, and contains fourteen valence electrons (four for carbon, six for oxygen, one each for the four hydrogens). Students should be able to construct a Lewis dot structure, making sure that the carbon and oxygen atoms have enough electrons to satisfy the octet rule (eight surrounding.